 
The nearly-free electron debacle showed researchers that any model that assumed that ions were in a sea of free electrons needed modification. This finding reduced many of the conclusions to examples of how a model can sometimes give a whole series of correct predictions, yet still be wrong. As soon as cyclotron resonance became available and the shape of the balloon could be determined, it was found that the assumption that the balloon was spherical did not hold, except perhaps in the case of caesium. This predicted a fairly large number of alloy compositions that were later observed. His idea was to add electrons to inflate the spherical Fermi-balloon inside the series of Brillouin-boxes and determine when a certain box would be full. Initially Hume-Rothery's attempts were quite successful. The nearly-free electron model was eagerly taken up by some researchers in this field, notably Hume-Rothery, in an attempt to explain why certain intermetallic alloys with certain compositions would form and others would not. Chemists generally steered away from anything that did not seem to follow Dalton's laws of multiple proportions and the problem was considered the domain of a different science, metallurgy. Despite all this progress, the nature of intermetallic compounds and alloys largely remained a mystery and their study was often merely empirical. The advent of X-ray diffraction and thermal analysis made it possible to study the structure of crystalline solids, including metals and their alloys and phase diagrams were developed. In the nearly-free model, box-like Brillouin zones are added to k-space by the periodic potential experienced from the (ionic) structure, thus mildly breaking the isotropy. In three-dimensional k-space, the set of points of the highest filled levels (the Fermi surface) should therefore be a sphere. In both models, the electrons are seen as a gas traveling through the structure of the solid with an energy that is essentially isotropic, in that it depends on the square of the magnitude, not the direction of the momentum vector k. With the advent of quantum mechanics, this picture was given a more formal interpretation in the form of the free electron model and its further extension, the nearly free electron model. A picture emerged of metals as positive ions held together by an ocean of negative electrons. With the advent of electrochemistry, it became clear that metals generally go into solution as positively charged ions, and the oxidation reactions of the metals became well understood in their electrochemical series. ( October 2009) ( Learn how and when to remove this template message)Īs chemistry developed into a science, it became clear that metals formed the majority of the periodic table of the elements, and great progress was made in the description of the salts that can be formed in reactions with acids. Unsourced material may be challenged and removed. 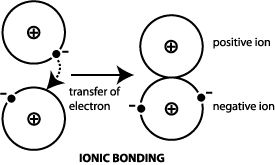
Please help improve this section by adding citations to reliable sources. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
